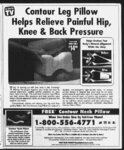
| Show CLARINEX® ki boffi Hie patients who withdrew pnmtiunly due to advene events was 2 CLARINEX and the placebo groups There wen no serious adverss events in these (desloratadine) trials in patients receiving desloratadine Afi advene events ttut wen reported by greater than or equal to 2 of patients who received the recommended daly doee of CLARINEX TABLETS Tablets (&0 mg oncedaly) and tost wen mom common wli CLARMEX than ptocabo an Med in Table 3 BfW Sunmuiy (Fir Ml PrttcriMi MOtCATKMSMH USAGE: hrfoniiitfiBf UU tw pKfcsfi Imirt) (XARINBiTaMelsareindlcaled Incidence ot Advene Events Reported by 2 2 d of Ptaienta In MuRipMose ClnicalTrtafe lor the relief of the nasal symptoms ol season? afieigic rttabs in patents 12 yew of ape end older end CLARINEX CONTRAMUCATHM: Tablets an contraindicaled in patents who ere' hypersensitive to this medication or to any of its ingredients or to taratadiiie PRECAunottfr CwwHmwrt BMutwrii hnpijninHt if rtfBfflf Mfonfr genic potential oF deslontadine was assessed using lorabeftie studies In an study in mice and a study in rad Madine ms administered in the diet at ddees DO YOU LEARN up to 40 mgkgday in mice (estimated desloratadine and deskntadkw matabotte exposures wen approximately 3 dmei the AUC in humane at the recommended daily onl dose) and 25 mglqyday in rats (estimated deskntadkie and deslontadine metabolite the recommended daily exposures wen approximately 30 times tie AUC in humans a ' onl dose) Male nice given 40 mgkgday lortiadine had a significantly higher incidena of hepaocefiular tumors (combined adenomas and carcinomas) than concurrent controls In rats a significantly higher incidence of hepatocellular turnon (combined nomas and carcinomas) was observed in males given 10 mgAgday and in males and females given 25 mgkday Hie estimated destontadbie ' during long-ter- use of deslontadine The hequency jnd magntude of laboratory and afepbocanfiognphic abnormaBBei wan sbniar in CLARINEX and placebiHretied patients Then wen no drfferences in advene events for subgroups of padents as defined by geKder age or The toGowring spontaneous advene events have been leportsd during the maikedng of destoratadkw tachycardia and nnlyhypenensfiMiynactions (such as nshpruri- tm urticaria edema dvsonaa and anaohvWsL and etevetod fiver anawnaa tadudbm mwi ' birabin and deslontadine metabolite exposures ofrats given 10 mgkg of loniadino wen approximately 7 times die AUC In humans at lbs recommended daily onl dose The cbnicaf significance of these findings ' is Mt known genotoxicity studies witti deslontaifine there was no evidence of genotadc potential in a reverse mutation assay (SamonME col mammalian mfcmsoma bacterial In mutagenicity assay) or in two assays for chromosomal aberrations (human peripheral blood lymphocyte ctastogenicity assy and mouse bone marrow minonuclous assy ) There was no effect on female 'fertility in rats at desloratadine doses up to piled on us nightly we’re up 'til 12 studying for things dial at that hour don't even make sens& In the morning we stumble into class sometimes unshowered Then is no krioimation to tndcato that abuse or and then the teacher complains Let’s think about this: We do homewoik but get nothing out of it—then we get into trouble phis we stink T? me there’s no benefit here-4t- atie RAartand V5 -- 0VERD08AGE: Intonnation nganlng acute ovsntosage is fmitod to experience hom dHal dials conducted during the development of tie CLARINEX producL In a dose ranging trial it doees of 10 mg and 20 mgAty somnolence was reported Skiole itaiv doses of 45 mo wan ahran to noimsl mala and (amah votontoen tar male specific decrease nfertHty demonstrated by reduced female conception rates decreased sperm numbers and motity and histopathologic testicular changes occurred at an oral desloratadine dose d( 12 mgkg in rats (estimated deslontadine wen manualy mad inabinderftashiqnbya subjects then was an increase in mean heart rale ol bpm rotative to placebo The QT interval was corrected for heart nSe (OTJ by both 10 dayi Al ECGs obtained in this study exposures wen approximately 45 txnes the AUC in humans ti the recommended defy onl dose) Desloratadine had no effect on fertility in rats at an onl dose of 3 mgtyday Pregaaecy Catag eey C: Deskntadm seas not teratogenic In nts at doses up to 48 mgkgday (estimated deslontadine and desioraladine metabolite exposures were' appradnkiely 210 times the AUC in humans (t the ncommended daly onl dose) or In rabbits at doses up to 60 mgAgday (estimated deslontadine exposun was approiid- -' matety 230 times the AUC in humans at the recommended daly oral dose) In a separate study an increase in loss and a decreased number of implantations and fetuses wen noted in female rats at 24 mgAg (estimated deslonta-(fin- e and deslontadine metaboHe exposures wen appmdmaiety 120 times the AUC in aX numans a i nt racofTHTwww my on oomj hboucnq oooy woioni ana now ngnong rtflex were npoiled in pups at doses of 9 mgAgttay or greater (estimated destaitia-din- e and destontafine metabolite exposure was approximately Slk times or greater Ian the AUC in humans at the recommended daily oral dose) Deslontadine had no effect pup deveiopmert at an onl dose o(3 mg4r(estimated desloratadine and deslontadine metabolite exposures were appnudnvrteiy 7 times ttie AUC in humans at the recommended daly onl dose) then an however no adequate and weficonjrolsd studies in pngnsnt women Because animal npnduction studies an not always predictive of human response deslontadine should be used during pregnancy on ty I AGUE AND DEPENDBICt DRUG dependency occun with CLARINEX Tablets 24 mgkgday (estimated desiontaifhe and desioraladine metaboHe exposures were in humans at the recommended approximately 130 times the AUC daly onl dote) A (estimated desloratadine and deslontadine metabotte exposures wars approximately 8 times the AUC in humans tithe recommended daly orti dose) Here ’t wkat iome teenagers said: When seven classes' worth of homework is (tardiotogisL In 92 CLARINEX-tnata- d the Bazett and Fridericia methods ' PrairiaVUIagKan Using the OTc(Baiett) then was a mean increase of I'll admit sometimes I 81 msec in CLARINEX-treate- d subjects rwladvwto ptacaba Using QTC (Frittarida) there was a mean inenasa of (L4 msec ki CLARINBC-treatasubjects nblive to niacabn No cSnicafiy relevant advene events wen raportad In the event of overdose consider standard measures to remove any anabsotbed drag Symptomatic and supportive treatment is ncommended Dssknttfns and at jTiyflngynwSMMiM Lemasiyoccurreo 11 U — ixx wnwaM complete homework just py nOTQoaiyiii Alllaon J mgmg or greater (esomanooeiiorau- -' ion aim in mntahniki irrpnsiiinii vnm appmnim11 it rats at one oosei 01230 irti iffihfitarwi nmih humans at the recommended daily onl dose) The oralmedan lethal dose in mice was deskntadkie exposures wen approxlmalely 290 times the 353 mgkg (estimated human daily oral dose on studying together —Atiiwon Lavty tik Wimwtta a mgm2 basis) No deaths occurred at oral doses up to 250 mgkg in monkeys (estimated dektontsdkie' Bqxrsures wen appratimsMy 810 times the human daily onl dose on a mg'm2 basis) - f ers explain something it's not enough I have to do it by myself at home —Wilbert Thomaat3 Palaoioa Tex All homework is busywork and a waste of time For me it’s more beneficial to talk to fiiends re lax and explore personal interests like lasers Jamie SeUbury 17 Randolph 8U account the importance of the drug to the mother MMrieUecThS safety and effectiveness of CLARINEX Tdbisls in pedtatcptfents under12 yean of age have not been established Geriatric Use CNnical studies of desioratatSno did not include sufficient numbert of subjects aged 65 and over to determine whether they respond dSwrendy from younger IMt MWMn 5uo6i um ijporteo ones cxpraioi ms nocRMnoMd onMncii In and selection for an dose eiderfy younger patients general tideriy patient should be m - ' reflecting the greater frequency of decreased hepsNc renal or cankac function and of concomitant disease or other drug therapy (see CUNICAL PHARSfiAPHHfiV Uriel rewhdim) cautious b tj ' IUVI YOU HAD A O! 55 Moauii timH OAKainr non f hdoiMta ' - AOVEMC REACtlOIIS: wpieef w trials 1838 patiwnts muKpte-do- e at doses of 2J mg to 20 mg daly K9patianls received the recommended daily dose of 5 mg In pafienls receiving 5 mg daly the rale ol adverse events was siritar between CLARINEX and ptacebrHreitad patients The percent of received CLARINEXTSMets In - b - ti a— —taydi wtaalVwta VclowT ewu raaiwrttiiiMwdatabFtta far MeataMentsshbukl be instructed to use CLARINEX TabHs as 1 Am — cm kpi nbuuqbo‘ arcqeaJ w biw w no woo wwoi on tii BoiKiBDUf CLARINEX Tablets may be taken wlhout regard to meals Metis should be advised not to increase the dose or dosing hequency as shakes' have not demonstrated increased effectiveness at higher doses and somnolsncs may occur pro ft Sometimes whien teach- tisariy needed Nwstag MeOwc Deslontaifine passes Mo breast nek thenfon a decision should be made whether tb discontinue nuising or to discontinue tfesloraladine taking into ' prior to class—and the knowledge I gain is slim But when Istudy at home for a quiz I find my parents’ enthusiasm is contagious and 1 enjoy the time we spend WHtwtwLyim Station NwtataltiC Sctwring Corporation KeniworthNJ 07038 USA MMM ' : 1201' U& Patent Nos :ImMi 28SK18ST-JB-S 459716 4JS3J31 4J04J6& Copyright0200lSchertngCoiponlktaAl 5907 and S100J274 righto nsafved I'- 1 Mfll ti MAHCH 10 SOOI NRAN MMA2MI |